2 Introduction
This chapter provides an overview of the subsurface behavior of DNAPLs and chlorinated-solvent plumes. Understanding these behaviors is crucial to a comprehensive understanding of any specific chlorinated-solvent site and therefore key to developing an accurate CSM, a critical component of the IDSS. A CSM describes the relationship of the source to the dissolved plume and other phases (e.g., vapor), including the site-specific transport mechanisms and receptor exposure pathways. The CSM is a framework for expressing site-specific knowledge that is then used in site management decisions. USEPA (in press) states that the life cycle of a CSM “mirrors the common progression of the environmental cleanup process where available information is used, or new information acquired, to support a change in focus for a project. The focus of a CSM may shift from characterization towards remedial technology evaluation and selection, and later, remedy optimization.”
Parallel to USEPA’s life-cycle CSM statement,
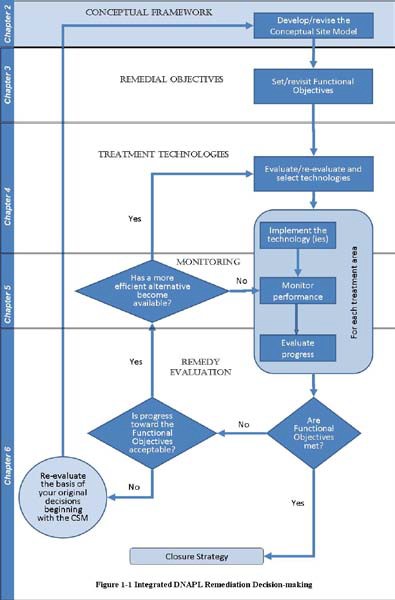
Figure 1-2 includes the CSM as the initial phase in development of an IDSS. CSM development begins when planning the site characterization, and the model is continually revised as additional information is generated during site remediation. The CSM is used during development of site remediation objectives and is updated during and after evaluation, selection, implementation, and monitoring of remediation technologies. Thus, the CSM supports development and evaluation of remediation objectives and treatment alternatives.
A common perspective is that source restoration focuses strictly on subsurface media that held or hold remnant nonaqueous liquid-phase mass—the “DNAPL source zone.” Groundwater restoration scientists and engineers now recognize the following: There is no bright line of demarcation between the “source zone” and the “diffuse plume.”Over time a large fraction of the chlorinated-solvent mass may migrate into the aquifer matrix, and this mass can still act as a reservoir of contaminants that sustains the plume.Many of the challenges of groundwater restoration at DNAPL sites arise as much from natural aquifer structure as from the characteristics of DNAPLs themselves.
Section 2.1 of this chapter discusses the elements of a CSM and its role in developing and implementing an IDSS. Section 2.2 describes the key processes involved in the movement, reaction, and attenuation of chlorinated solvents in the subsurface. Section 2.3 highlights the importance of geologic conditions that control subsurface transport and, thus, many remediation processes. Section 2.4 describes issues pertaining to plume dynamics over time, including plume growth/attenuation and subsurface responses to treatment across different media and contaminant phases.
Section 2.5 presents a tool, the 14-Compartment Model (Sale and Newell 2011), which provides a framework to track chlorinated-solvent mass in the subsurface and how its distribution within subsurface compartments relates to treatment goals, remediation alternative selection and design, and site monitoring. Section 2.6 presents an example of a chlorinated-solvent site and development of its CSM. (This site is used in subsequent chapters to illustrate other elements of an IDSS and decision making.)
This overview of the behavior of DNAPLs and chlorinated-solvent plumes in the subsurface highlights key issues in development of a CSM. In-depth treatment of all technical issues is not provided. Instead, the goal of this chapter is to identify the key technical issues to be considered when developing a CSM. Additional literature citations are provided where appropriate, including previous ITRC documents that address pertinent technical and regulatory considerations.
2.1 The Conceptual Site Model
Conceptual Site Model A CSM is a framework for site- specific knowledge that can guide site management decisions. The CSM describes the site, including the relationship of the source to the dissolved plume and other impacted phases (e.g., vapor) and site-specific transport mechanisms and receptor exposure pathways.
A CSM is a comprehensive description of a site used to support characterization or remediation planning and implementation. While there are a number of definitions for a CSM, it generally describes the relationship of the source to the plume and other affected media and includes contaminant fate and transport mechanisms and potential exposure pathways (ITRC 2004b). The CSM can also incorporate other societal or regulatory considerations, such as land use, community plans, and risk management scenarios. The CSM is a powerful, useful tool for project
decision makers. It is often presented using a variety of media, including text, maps, cross sections, two- or three-dimensional graphics, physical or mathematical models, tables, charts, and other visual representations. The CSM is a working hypothesis and should be tested and revised as new site data are gathered.
The CSM is especially important for chlorinated solvent–contaminated sites because at any given time chlorinated solvents are typically distributed across multiple geologic media and occur in more than one chemical phase in the site subsurface. Movement of contaminant mass between these phase-media “compartments” impacts overall contaminant transport at a site (Pankow and Cherry 1996, Reynolds and Kueper 2001) and the response of one compartment to treatment of another (Chapman and Parker 2005; Parker, Chapman, and Guilbeault 2008; Kavanaugh et al. 2003; Stroo et al. 2003; Falta, Rao, and Basu 2005). These interrelationships also impact the efficacy of remediation efforts and ultimately determine the success of a remediation approach. For example, if source treatment is used to reduce plume concentrations, then the relationships between a source and its plume, and the expected response of both to treatment, must be understood. Similarly, if vapor impacts to indoor air are to be eliminated via groundwater plume remediation, then the relationship between groundwater, soil gas, and indoor air volatile organic compound (VOC) concentrations, and the response in these media and phases to groundwater treatment, must be understood as part of the CSM.
In summary, the CSM must be based on the site geologic and hydrogeologic setting as well as the physical and chemical properties of the contaminants, the site geochemical conditions, and the relevant transport and attenuation mechanisms within and between phase-media compartments. The CSM must describe all media that are impacted, including the vadose and saturated zones, and geologically and anthropogenically controlled heterogeneities, as well as multiple chemical phases (i.e., DNAPL, vapor, dissolved, or sorbed) that may be present.
2.2 Key Subsurface Transport and Reaction Processes
This section is an overview of the key transport and reaction processes that occur during and after subsurface releases of chlorinated solvents. Pankow and Cherry (1996) present a more detailed review of chlorinated-solvent transport and occurrence.
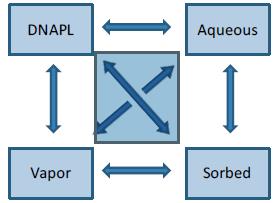
Chlorinated solvents are usually released in liquid phase (DNAPL) though in some cases an aqueous solution containing dissolved VOCs may be released. Once in the subsurface, the chlorinated solvents can exist in four phases: DNAPL, aqueous, vapor, or adsorbed. The adsorbed phase is normally associated with organic carbon, although other forces can contribute to soil adsorption. Mass transfer among the four chemical phases (Figure 2-1) distributes the chlorinated-solvent mass in accordance with equilibrium constants, although each rate can be modified by multiple chemical and physical processes (Mackay 1991). Diffusion limitations on interphase mass transfer are particularly important because these can significantly slow mass transfer of chlorinated solvents from the DNAPL phase into the aqueous, vapor, and sorbed phases. Diffusion also controls the rate of mass attenuation in the later life-cycle stages of a release. Diffusion occurs at the soil pore scale over distances of a few millimeters in a single geologic media, as well as at larger scales between different domains defined by geologic heterogeneities. The effects of geologic heterogeneity are discussed in greater detail below.
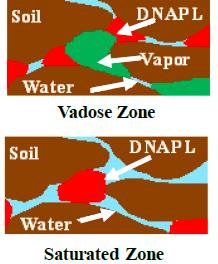
In the vadose zone the vapor phase is significant in terms of mass storage and transport, while in the saturated zone the vapor phase is essentially absent under most conditions (Figures 2-2 and 2-3). Capillary forces interacting with the aquifer matrix control the movement and distribution of the DNAPL in the subsurface. Most soils, sediments, and bedrock matrices are typically water-wet in the saturated zone, which means that water occupies the smallest pore spaces and preferentially coats the subsurface matrix. DNAPL is generally nonwetting with respect to the aqueous phase, which means that DNAPL has to overcome the capillary retention of water in the pore spaces to migrate through the saturated zone. The direction of migration is controlled by the mass of the DNAPL column and the size of the pore spaces, with preferential flow in the larger pore spaces that have lower capillary forces for the DNAPL to overcome. These migration pathways can be very tortuous due to heterogeneities in the subsurface. Though denser than water, DNAPLs may migrate horizontally or in other directions than expected due to the size and distribution of preferential pathways.
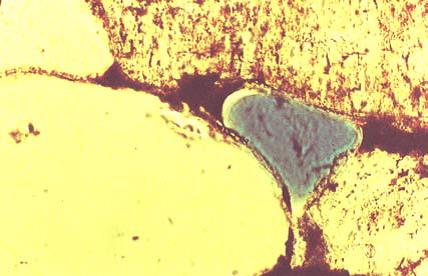
Saturated Zone figure 2-2. Pore-scale distribution of chemical phases of chlorinated solvents(On the right)
Figure 2-3. Photograph of nonaqueous- phase liquid and water sharing pore space in sand. Source: Wilson et al. 1990 (On the left)
Reactions that degrade or transform chlorinated solvents are very important to the subsurface fate and transport of chlorinated solvents. Naturally occurring reactions include microbial degradation and chemical degradation reactions. The most common attenuation reaction for chlorinated ethene is reductive dechlorination (ITRC 2008b), where under the proper reducing conditions, tetrachloroethene (or perchloroethene [PCE]) is reduced to trichloroethene (TCE), cis-1,2-dichloroethene (cDCE), vinyl chloride (VC), and ethene. The reader should be keenly aware that the degradation of a particular compound can significantly slow down as the reaction proceeds or the biological reductive dechlorination sequence can be interrupted by oxidative biological processes or abiotic degradation mechanisms.
The above processes interact to control the fate and transport of chlorinated solvents following a subsurface release. The following assumes the initial release is in the form of a DNAPL. Initially, DNAPL displaces air and/or water from the pore spaces. Then, as gravity moves DNAPL downward, it can leave a trail of residual blobs or ganglia of DNAPL, held in place by capillary forces.
When released in sufficient volumes, DNAPL can also form continuous bodies referred to as “pools.” As shown in Figure 2-4, DNAPL pools may occur on top of low-permeability layers. Where DNAPL is present as a mixture of compounds, individual constituents of the DNAPL dissolve into water, sorb to solids, and partition into soil gas at different rates based on their physical and chemical properties and the percentage of each compound in the DNAPL. This fact reinforces the point that managing chlorinated solvents is not just about managing DNAPL. It is also about managing DNAPL constituents dissolved in water, sorbed to solids, and volatilized into soil gas.
2.3 The Importance of Geologic Conditions
Further insight into the key subsurface transport processes affecting a chlorinated- solvent release is gained by recognizing the importance of the heterogeneous geologic conditions, as shown in Figure 2-4. There are two main consequences of heterogeneity: the location and distribution of DNAPL (DNAPL architecture) and diffusion of dissolved solvents from relatively transmissive zones to low- permeability zones.
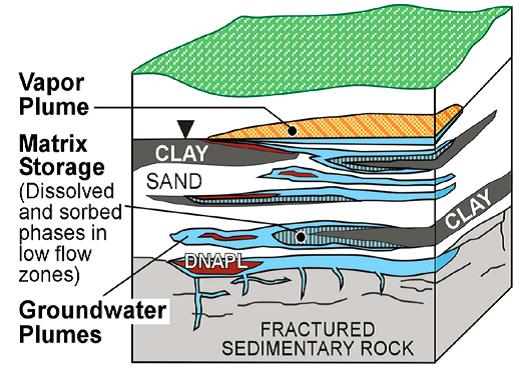
Figure 2-4. DNAPL pools, groundwater, and vapor plumes and matrix storage in a heterogeneous setting. Source: Sale et al. 2008.
DNAPLs preferentially move through zones with the greatest permeability and transmissivity. The highest permeability and most transmissive portions of the subsurface matrix may be very small and take up only a fraction of the aquifer matrix. In low-permeability portions of the subsurface, DNAPL can be largely precluded from entering these areas by capillary forces. An important exception occurs where secondary features such as fractures, root casts, or animal borings are present in low-permeability layers. In the end, subsurface DNAPL occurrence is often conceptualized as sparsely distributed fingers of DNAPL ganglia and pools. Intervals where DNAPL is present are surrounded by intervals that are largely free of DNAPL. The sparse distribution of DNAPL partially explains why it is difficult to locate during characterization work.
Text Box 2-1. Air Force Plant 44, Diffusive Storage (see Appendix A)
A new CSM indicated that even though soil vapor extraction (SVE) had effectively removed TCE from the vadose zone, chlorinated-solvent contamination was still discharging to the downgradient aquifer due to diffuse storage in fine-grained materials in the capillary fringe and in the upper part of the aquifer material. SVE did not effectively remove this material.
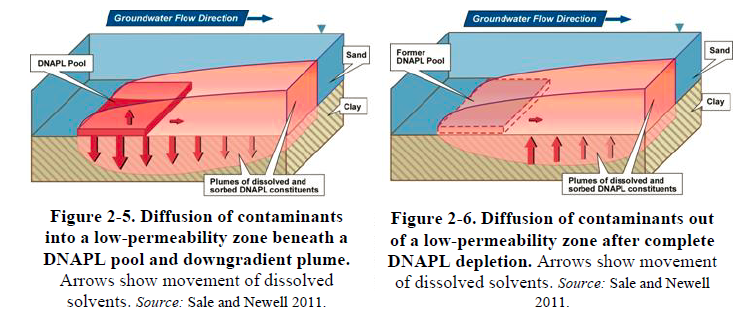
Another consequence of preferential pathways is that high concentrations of dissolved contaminants in the transmissive zones drive contaminants into low-permeability zones via diffusion. Within low-permeability saturated zones, contaminants are stored as an aqueous phase in water and as a sorbed phase on or in solids. The process of contaminants moving into low- permeability layers via diffusion is referred to as “matrix diffusion.” The significance of contaminants in low-permeability portions of the matrix is that they diffuse back (back-diffuse) into the higher transmissive zones once concentrations in the latter decrease (see Text Box 2-1). Thus, back-diffusion can sustain dissolved plumes in transmissive zones long after the DNAPL source is gone (e.g., Chapman and Parker 2005; AFCEE 2007; Payne, Quinnan, and Potter 2008; Sale et al. 2008). However, the degree to which contaminants diffuse into low- permeability layers and are later released varies with the volume and concentration of the original source, distance from the source, time from release, and relative permeability of the subsurface strata. Figures 2-5 and 2-6 illustrate the process by which contaminants are stored and released from low-permeability zones.
2.3.1 Geologic Heterogeneity
Groundwater contamination first captured national attention in the 1960s and 1970s. At that time, environmental restoration practitioners did not realize the importance of aquifer matrix heterogeneity as a primary factor controlling contaminant migration. Even today, CSMs are often based on an assumption of the plume spreading within a uniform homogeneous aquifer.
Many traditional hydrogeologic tools average subsurface properties over large scales (hundreds or thousands of yards/meters or miles/kilometers). Aquifer pumping tests are typically analyzed with solutions, such as the Theis equation, that assume homogeneous, isotropic conditions and result in volume-averaged hydraulic properties. While this information is relevant for evaluating well productivity, the limitations of neglecting heterogeneity when evaluating solute transport were recognized by Theis (1967) himself: “I consider it certain that we need a new conceptual model, containing the known heterogeneities of natural aquifers, to explain the phenomenon of transport in groundwater.” The frequent neglect of geologic heterogeneity persists to this day, as more recently stated by de Marsily et al. (2005): “Hydrogeology has been too much inclined toward hydraulics and solving of the flow equations, and not enough toward geology and understanding/describing the rock structure, facies, and properties in a geologically realistic manner, thus proposing ‘exact’ solutions, but to poorly posed problems.”
Unfortunately, traditional hydrogeologic tools do not accurately represent or account for the importance of geologic heterogeneity within a single plume or even on a single contaminated site. The oversimplification of subsurface geologic conditions leads to the elliptical contaminant isoconcentration lines often drawn on plan view maps or in hydrogeologic cross sections. In reality, plumes migrate primarily in the most transmissive portions of an aquifer, which are surrounded by lower-permeability media that store and release contaminants via diffusion. This more refined and realistic conceptual model of contaminant transport has been supported by natural gradient tracer studies (Mackay et al. 1986; LeBlanc et al. 1991; Rivett, Feenstra, and Cherry 2001; Payne, Quinnan, and Potter 2008) and in recent years by high-resolution characterization of groundwater plumes resulting from chlorinated-solvent source zones (Guilbeault, Parker, and Cherry 2005; Einarson and Mackay 2001), which has led to several observations:
- Order of magnitude changes in groundwater contaminant concentrations occur over short distances (often a meter or less from a source) transverse to flow.
- Chlorinated-solvent concentrations in the cores of plumes, at large distances downgradient of source zones, are often similar to concentrations in source zones.
- Transmission of the vast majority of plume mass can occur through a very small portion of the subsurface.
These observations have led to the realization that hydrodynamic dispersion of contaminants is likely a weak process in the absence of geologic heterogeneity and that geologic heterogeneity is the dominant control over contaminant plume spreading.
At scales of 1–100 m, subsurface heterogeneities and anisotropies lead to complex groundwater flow and contaminant mass transport patterns, especially for chlorinated solvents where DNAPL transport, interphase mass transfer, and diffusion are each strongly controlled by subsurface heterogeneity and together control the development of groundwater plumes. Field tracer studies (Mercado and Halvey 1966; Mackay et al. 1986; LeBlanc et al. 1991; Hall, Luttrell, and Cronin 1991; Stephens et al. 1998, Payne, Quinnan, and Potter 2008) have shown that a relatively small portion of most aquifers having the greatest permeability form preferential pathways where the highest groundwater and contaminant flows occur and where the actual transport velocities are much greater than aquifer-wide estimates made using traditional, large-scale averaging methods that assume homogeneous conditions. This understanding of subsurface transport processes suggests that the common approach of averaging out heterogeneities and representing the subsurface as homogeneous inadequately represents the subsurface and makes it more difficult to predict contaminant migration and restore groundwater quality to acceptable levels.
Many common hydrogeologic tools can underestimate or incorrectly represent the effects of geologic heterogeneity. Analytical contaminant transport models that assume homogenous conditions essentially average flow and transport processes throughout the plume and, therefore, understate maximum transport velocities in heterogeneous media. Further, numerical models are often discretized with a low level of geologic detail due to a lack of high-resolution site characterization data. This limitation affects plume flow and transport estimates in the same manner as seen with analytical models. Analytical or numerical models that underrepresent heterogeneity must instead explain documented plume spreading using unrealistic dispersivity terms. Overall, preferential flow and transport in the heterogeneous conductive zone are often underestimated (underrepresented), and lateral plume spreading is often overestimated.
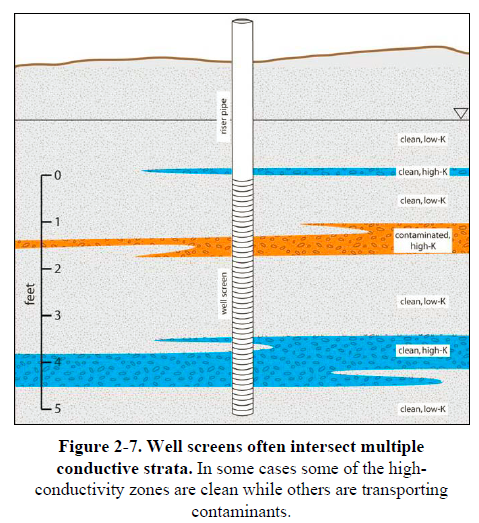
Substantial literature exists that describes porous media flow processes and modeling in the presence of multiscale heterogeneities. Upscaling techniques and other approaches to more accurately predict flow and transport in multiscale heterogeneous media also exist (Frippiat and Holeyman 2008), but detailed characterization data to enable their use are lacking at many contaminated sites. Even where mathematical upscaling techniques are used to represent effects of multiscale heterogeneities in flow models, this approach may not pinpoint the high-flow zones at a site that may transmit most of the contaminant flux. Fully penetrating monitoring wells often provide an incomplete representation of site conditions (Figure 2-7). Wells with long screened intervals average results
over the screen interval and likely do not detect high-concentration
and high–mass flux zones that are limited in vertical extent. As a
result, fully penetrating monitoring wells may under represent maximum
contaminant concentrations by an order of magnitude or more. Where warranted by project goals, it is beneficial to characterize aquifer structure at a resolution sufficient to properly account for subsurface heterogeneity and to physically identify key heterogeneities dominating contaminant
transport. Such an approach produces an understanding of site Hydrogeology sufficiently detailed to support development of a realistic CSM and a successful IDSS. Depth-discrete VOC sampling and/or relatively high-density grids or transects are the most obvious of high-resolution characterization data. Ideally, high-resolution characterization of subsurface geologic conditions and hydraulic properties should be conducted. The higher-resolution characterization approach also improves understanding of contaminant mass flux (Guilbeault, Parker, and Cherry 2005; ITRC 2010b).
2.3.2 Aquifer Structure—A Multiporosity System
The structure of preferential-flow regions at heterogeneous sites and their interaction with less- conductive zones often controls contaminant distribution and transport. If the subsurface is actually heterogeneous but is treated as if it were homogeneous, then a very small effective porosity must be used to explain the actual transport velocities. However, since porosity is a property of a representative elementary volume of the porous media, using it as a lumped parameter (averaged over a large volume of heterogeneous media) ultimately misrepresents the actual subsurface transport process (Zheng and Bennett 1995).
Most if not all subsurface environments can be thought of as having mobile and immobile (i.e., markedly less mobile) porosity domains, which impact or even control contaminant transport. The concepts of dual-porosity and dual-permeability domains have been used to conceptualize and model mobile and immobile systems in fractured and highly heterogeneous subsurface environments (Warren and Root 1963; van Genuchten 1985; Berkowitz, Bear, and Braester 1988). Dual-porosity and dual-permeability flow models essentially overlay the mobile and immobile media within a single element of the subsurface. Dual-porosity models divide the total porosity within each element between a mobile porosity that transmits flow and an immobile porosity that is relatively stagnant. In a dual-permeability model, both media within each element can transmit flow with different assigned hydraulic conductivity values. Both dual- porosity and dual-permeability models simulate diffusive transport between the mobile and immobile media within each element. A number of mathematical vadose zone and groundwater flow and transport models incorporate dual-domain (i.e., dual-porosity or dual-permeability) representations, including MT3DMS (Zheng and Wang 1999), MODFLOW-2005 CFP (Shoemaker et al. 2008), MODFLOW SURFACT (HydroGeoLogic 1998), Hydrus (Simunek, Sejna, and van Genuchten 1999), and PHREEQC (Parkhurst and Appelo 1999). Dual-domain flow and transport models still represent a simplification of real-world conditions and therefore have inherent limitations. Nonetheless, where specific conditions and objectives warrant, they may represent an improvement over traditional, single-domain modeling approaches.
An alternative to the dual-domain approach is the streamtube approach, where flow characteristics in individual streamtubes are varied to represent flow in heterogeneous media. Streamtube flow models have been used to conceptualize heterogeneous flow and to simulate experimental data, primarily to evaluate petroleum production (Lenormand 1995, Lenormand and Wang 1995, Emanuel and Milliken 1997). Streamtube flow models have not been incorporated into groundwater flow simulators as have dual-domain models except at a research level (Finkel, Liedl, and Teutsch 1998).
2.3.3 Bedrock Systems
In summary, the use of single-media or single-porosity concepts to characterize heterogeneous systems mandates the use of a very small effective porosity which, in turn, underrepresents the profound influence of heterogeneity on transport. The value of effective porosity is also often overestimated using specific yield or other parameters that are not representative of heterogeneous transport conditions. This practice can lead to a dramatic underestimation of actual transport velocities in the mobile portions of the aquifer. On this basis, Payne, Quinnan, and Potter (2008) suggested that the terms “mobile porosity” and “immobile porosity” are more appropriate than the terms “total porosity” and “effective porosity” to describe heterogeneous subsurface systems The dual-porosity concept may represent an improvement in the representation of highly heterogeneous media although the absolutely explicit prediction of contaminant transport and plume evolution is still a highly elusive goal. The dual-porosity approach, an increase in the use of higher-resolution site characterization methods, and the incorporation of more small-scale heterogeneities in transport models are all more explicitly accurate approaches to representation of chlorinated-solvent plumes. Depending on project goals, these more advanced methods may be justified to support development of a CSM and an IDSS.
Groundwater flow and contaminant transport in bedrock systems are typically dominated by secondary fracture porosity with matrix diffusion of contaminants into and out of primary porosity significantly affecting contaminant flow and distribution in the aquifer (Parker, Gillham, and Cherry 1994). The dual-domain, multiporosity model used to describe flow in unconsolidated materials as discussed above can also describe contaminant transport and groundwater flow in bedrock. The porosity available for movement of fluids in bedrock is commonly subdivided into three classes (Sowers 1981):
- Primary porosity—the openings between individual grains of rock, which can range from negligible (metamorphic rock) to high (sandstone). The rate of matrix diffusion is a function of primary porosity.
- Genetic porosity—the openings associated with specific depositional materials or environments, examples of which include lava tubes, tufa tubes, hollow-grained volcanic ash, and diatomaceous limestone.
- Secondary porosity—the openings or discontinuities in a rock matrix caused by breakage, fracture, or dissolution, which are further subdivided by origin as faults, joints, or karst channels.
Secondary porosity is frequently the dominant pathway for rapid fluid flow in fractured rock, while primary and genetic porosity play an important role in storage or retention of groundwater contamination. The fractures typically occupy a very small fraction of the aquifer matrix volume, but the hydraulic conductivity of the fracture zone is normally quite high. As a result, fractured bedrock can form low-water-volume, high-transport-velocity aquifers. Payne, Quinnan, and Potter (2008), for example, report natural gradient tracer studies in fractured sandstone where the tracer migrated more than 12 m per day. When slug and pumping tests in this aquifer were analyzed using calculation tools designed for homogeneous, isotropic porous media, the estimated average groundwater velocity for the formation was less than 3% of the observed transport velocity. Observations such as these call attention to the importance of aquifer structure in groundwater flow and solute transport.
The occurrence and nature of porosity and groundwater flow paths are exceedingly complex, and there is much research under way on methods of characterization, modeling, and prediction. The subject resists quick summation on all but the most superficial level. Readers are encouraged to explore further information available on the Internet, such as “Multidisciplinary Characterization of Contaminant Transport in Fractured Rock, Mirror Lake, New Hampshire” at http://toxics.usgs.gov/sites/mirror_page.html (USGS n.d.).
The multiple porosity and highly heterogeneous nature of fractured rocks requires an extension of current contaminant flow theory and field methods. It is particularly important that theoretical developments be applicable on a scale commensurate with field measurements.
2.4 Chlorinated-Solvent Source and Plume Dynamics
This section describes how chlorinated-solvent plumes form and evolve, as well as how they attenuate and respond to treatment.
2.4.1 Maturation of Subsurface Chlorinated-Solvent Releases
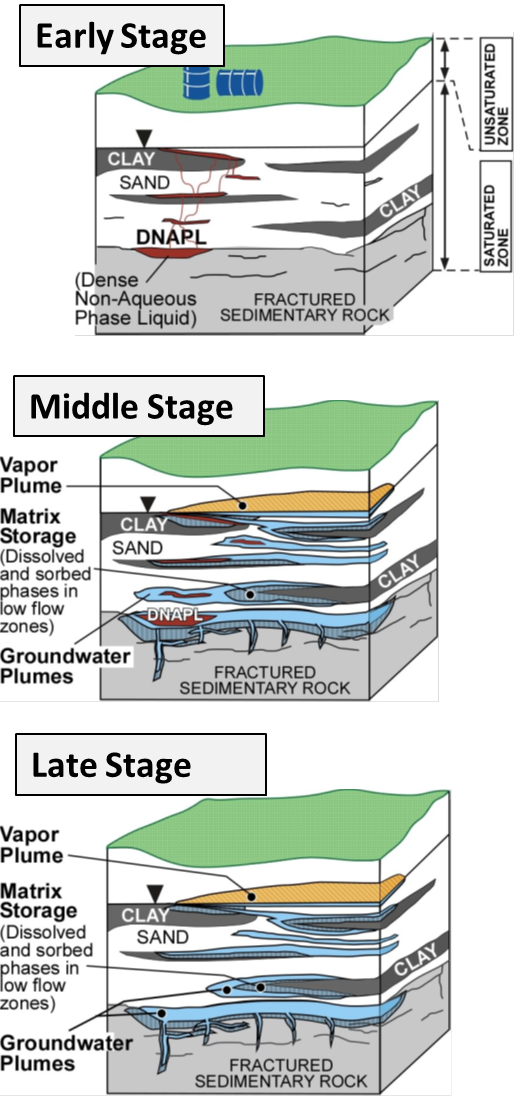
It is essential to remember that chlorinated-solvent releases continue to evolve (Figure 2-8). Immediately after a chlorinated-solvent release, the source area is dominated by DNAPL. With time DNAPL dissolves, and plumes develop while contaminants accumulate in low-permeability zones. Ultimately, little or no DNAPL remains, and plumes are sustained by the release of contaminants from low-permeability zones via diffusion (back-diffusion) (Chapman and Parker 2005). Although recoverable DNAPL may be found within some source zones, it is notoriously difficult to find DNAPL. At some sites (see late stage in Figure 2-8), it simply may not be there any longer, even though the source zone still contributes contaminant mass to the downgradient plume. Key factors controlling the rate at which chlorinated- solvent releases evolve include the amount of chlorinated solvent released, effective solubility of the DNAPL constituents, rate of groundwater flow, and the architecture of the relative permeabilities. Critically, at some sites impacted by chlorinated solvents, DNAPL may never have been present, for example, a site where wastewater with high aqueous concentrations of chlorinated solvents was stored in an unlined pond.
2.4.2 Source and Plume Response to Treatment
This section discusses several key processes involved in the subsurface response to treatment, including the impact of source treatment on plume mass flux and concentration and the response of chlorinated-solvent concentrations in soil vapor and indoor air to groundwater plume treatment. These topics illustrate the importance of a CSM that realistically represents the interactions between chlorinated-solvent mass in different subsurface compartments.
Figure 2-8. Multiple stages in the evolution of a chlorinated-solvent release site. Source: Sale et al. 2008.
The relationship between DNAPL mass removal and reductions in mass flux from sources zones is a subject of ongoing research. See Use and Measurement of Mass Flux and Mass Discharge (ITRC 2010b) for a detailed discussion of mass flux and mass discharge measurements and applications.
While it might be assumed that there is a one-to-one relationship between DNAPL mass removal and mass flux reduction, the DNAPL architecture and heterogeneity at a particular site influence this relationship. Published studies have found that in some cases (e.g., when DNAPL is isolated from the high-permeability flow paths) mass flux reductions greater than the degree of DNAPL source removal have been reported. In instances where the subsurface is more homogeneous, mass flux was reduced less than the extent of DNAPL source removal (Brusseau et al. 2008, DiFilippo and Brusseau 2008).
Research has shown that geologic heterogeneity and DNAPL distribution/architecture are key factors that control the relationship between source mass depletion and reduction of plume flux (Falta, Rao, and Basu 2005; Jawitz et al. 2005). Basu, Fure, and Jawitz (2008) compared the results of complex numerical simulations using the UTCHEM simulator to simplified analytical models of source depletion (e.g., Falta, Rao, and Basu 2005 and others) in terms of their ability to accurately predict the relationships between source mass depletion and plume flux reduction. They showed that the simplified analytical models provided a reasonably accurate estimate of the source-plume relationship, as indicated in Figure 2-9, which illustrates the following key concepts:
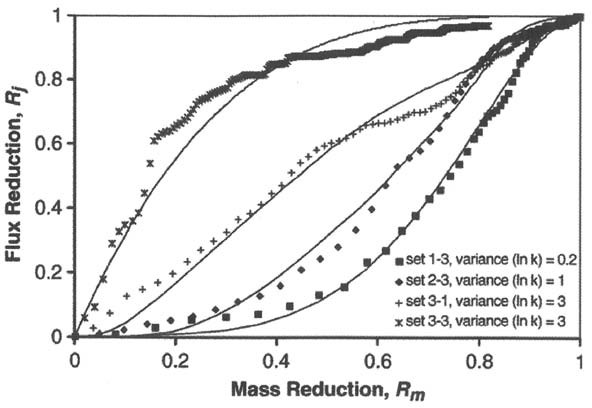
Figure 2-9. Comparison of modeled relationships between source mass depletion reduction and predicted plume flux reduction obtained using UTCHEM-generated simulations (symbols) and simplified analytical solutions (solid lines) for a range of geologic heterogeneity conditions represented by the log of hydraulic conductivity variance.
Source: Basu, Fure, and Jawitz 2008.
- The simplified analytical solutions (lines in Figure 2-9) closely approximate the UTCHEM numerical simulations (symbols in Figure 2-9).
- The source mass flux reduction and the plume flux reduction do not follow a 1:1 linear relationship for most site conditions.
- For heterogeneous sites (large variance in Figure 2-9), a small fraction of source mass reduction results in a relatively greater plume flux reduction, reflecting that the plume flux is controlled predominately by the higher-permeability zones, which are where source treatment preferentially occurs.
- For homogeneous sites (small variance in Figure 2-9), a large fraction of source mass reduction results in a relatively smaller plume flux reduction, reflecting that in the absence of geologic heterogeneity, a partial reduction in DNAPL mass does not significantly decrease the DNAPL dissolution rate into flowing groundwater.
The primary factor affecting the different curves in Figure 2-9 is heterogeneity, expressed as the variance in the natural log (ln) of the hydraulic conductivity (K) in the various geologic media at the site. Other factors that control the source-plume relationship following partial source treatment include the specific DNAPL geometry with respect to heterogeneity and other parameters such as the DNAPL ganglia-to-pool ratio and other geometric factors. Numerical simulators such as the UTCHEM code explicitly account for these factors, while the simplified analytical models use a lumped fitting parameter (e.g., “gamma” in Falta, Basu, and Rao 2005) to account for these factors. Basu, Fure, and Jawitz (2008) developed a regression relationship between the variance in ln K and the model-fitting parameter gamma used by Falta, Basu, and Rao (2005), which can be used to estimate a value of gamma for a site.
In summary, understanding source and plume response to remediation is a key question at many sites. Site managers ask questions such as, “Will source remediation alone meet site goals?”, “What will happen if no action is taken?”, “Should I combine source and plume remediation?”, and “What is a reasonable remediation objective?” One powerful but relatively simple tool that can be used to help with these questions is USEPA’s REMChlor model (Falta, Rao, and Basu 2005; Falta et al. 2006). REMChlor combines a source zone model based on the gamma function above with an advection-dispersion model for chlorinated solvents. Two strengths of REMChlor are that is it can simulate how a source responds to remediation without the need for high- resolution source zone architecture data and that it can show, in a general way, how the plume will respond to partial source zone and/or plume remediation. Weaknesses are that, while it can simulate matrix diffusion effects in the source zone, it does not account for matrix diffusion effects in the plume downgradient of the source, and it has the theoretical disadvantages of the advection-dispersion approach (see Section 5.3 for a more detailed discussion). Overall however, both researchers and practitioners have found REMChlor to be a relatively simple but powerful groundwater model for evaluating remediation at chlorinated-solvent sites.
2.4.3 Vapor Intrusion
Management of potential risks associated with vapor intrusion is increasingly becoming a regulatory driver for site cleanups. The most basic instance of risk associated with vapor intrusion is derived from contaminated vadose zone soils, while in other instances volatilization of contaminants from groundwater is associated with vapor intrusion (ITRC, 2007; USEPA, 2015; Ma et al., 2020). Although this document is not largely focused on vadose zone impacts, their management plays an integral role in developing an integrated strategy for managing sites with DNAPLs. In some cases, regulatory agencies and responsible parties have negotiated (or are considering) site-specific groundwater cleanup criteria selected to be protective of indoor air in buildings above or near the groundwater plume.
Figure 2-10 shows an example from USEPA of a CSM cross section for potential subsurface vapor intrusion pathways (USEPA 2002, ITRC 2007c). The figure illustrates that the saturated groundwater zone represents only a portion of the overall mass of chlorinated solvents in the system and that the capillary fringe forms the actual boundary condition for soil vapor migration into the vadose zone. The transient processes following in situ groundwater treatment that controls the attenuation of soil gas concentrations of organic vapors includes a combination of interphase mass transfer, desorption, diffusion, and advection. While the processes involved in soil gas organic vapor attenuation following in situ groundwater treatment have not been previously reported in the published literature, it is expected that some period of time will be required before the chlorinated-solvent mass in the vadose zone attenuates to a new chemical equilibrium with the decreased groundwater concentrations,
especially if the vadose zone is highly heterogeneous and contains a reservoir of chlorinated-solvent mass in lower-permeability regions
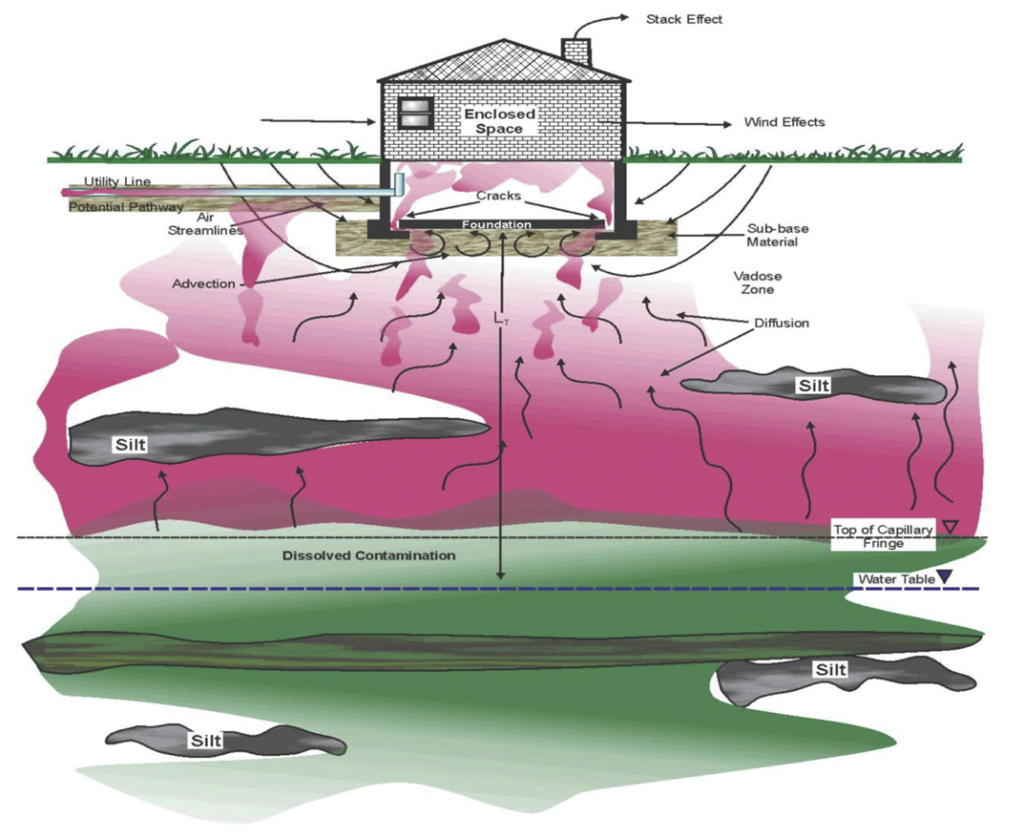
Figure 2-10. Conceptual model for subsurface vapor pathways. Source: USEPA 2002.
2.5 Introduction to the 14-Compartment Model
The 14-Compartment Model (Sale and Newell 2011) is used in this document to illustrate a number of key concepts. The following sections introduce the 14-Compartment Model and illustrate its application to assess the stages of plume maturity and to represent conditions at an example site. Subsequent chapters of this volume make use of the 14-Compartment Model to illustrate relevant concepts related to developing and implementing an IDSS (Note an online version of the 14-compartment model is available (Marquardt et al., 2014); search for “14-Compartment Model” “Online” “ESTCP.”).
2.5.1 The 14-Compartment Model
The 14-Compartment Model is a tool to aid in developing a simplified conceptualization of subsurface releases of chlorinated solvents. It is used to represent conditions in the saturated zone, and although the concepts could be applied to conceptualize contaminant impacts in the vadose zone, this step is not undertaken in this document. The 14-Compartment Model provides a holistic view of chlorinated-solvent phases within the saturated zone and their distribution in transmissive and low-permeability zones in source zones and plumes. The model also assists in the expected compartment response to treatment. Finally, the 14-Compartment Model can be used to assist in setting site remediation goals and to evaluate remediation alternatives and site monitoring data. It may not be necessary to numerically characterize all fluxes between compartments.
The 14-Compartment Model is not a numerical or analytical model—instead, it provides an organized framework to assess contaminant mass transfers between the source zone and plume and between relevant compartments within the source and plume regions. It provides a screening-level analysis of the benefits of potential remedial measures. The following are highlights of the model:
- The 14-Compartment Model was developed with a focus on mass transfers between compartments, not on static mass storage. Although the data used to assess mass transfers could also be used to approximate contaminant mass in storage, the model is focused on mass transfers to best support remedy selection and design.
- Contamination in each compartment is represented by a qualitative gauging of the chemical potential, which has been expressed as an “aqueous-phase-equivalent concentration.” By using chemical potential (or its surrogate), we can see whether diffusive transfers are occurring and in which direction they are going.
- Mass transfers can be advective or diffusive. Transport in transmissive zones is primarily advective, while transport within lower-K zones (and between lower-K and transmissive zones) is dominated by diffusion.
- Advective transfers are irreversible; diffusive transfers are reversible, with the exception of diffusion-controlled dissolution of DNAPL.
- Transfers between low- and higher-K zones are predominantly diffusive.
- Diffusive transfers flow from compartments with higher chemical potential to compartments with lower chemical potential.
- As a source becomes exhausted and plume structure evolves, compartments that were net recipients of contaminants early in the source/plume development can become net sources of mass transfer late in the plume evolution process. The problem evolves from one of contaminants in transmissive zones to one of contaminants in lower-permeability zones through time.
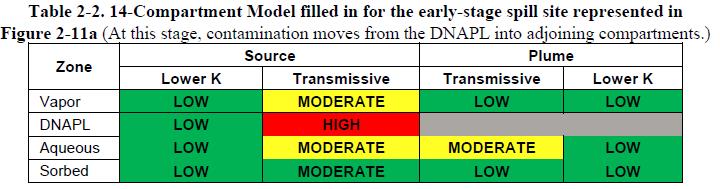
Chlorinated solvents can occur in four phases in the source zone (DNAPL, aqueous, sorbed, and vapor) and three phases in the plumes (following NRC 2005, there is no DNAPL in plumes). Each of these phases can occur in subsurface zones that can be classified as “transmissive” (mobile) or “lower permeability” (immobile). Figure 2-11a provides a key to the locations of each of the 14 compartments at a hypothetical spill site. DNAPL has entered the subsurface, and there is DNAPL in the vadose zone as well as the aquifer. The aquifer comprises sand (transmissive) and clay (lower-permeability) zones. The source zone is defined as that portion of the site that has (or once had) direct contact with the DNAPL, and the plume is the portion of the site downgradient from the DNAPL zone that will receive contaminants via dissolved-phase advective transport.
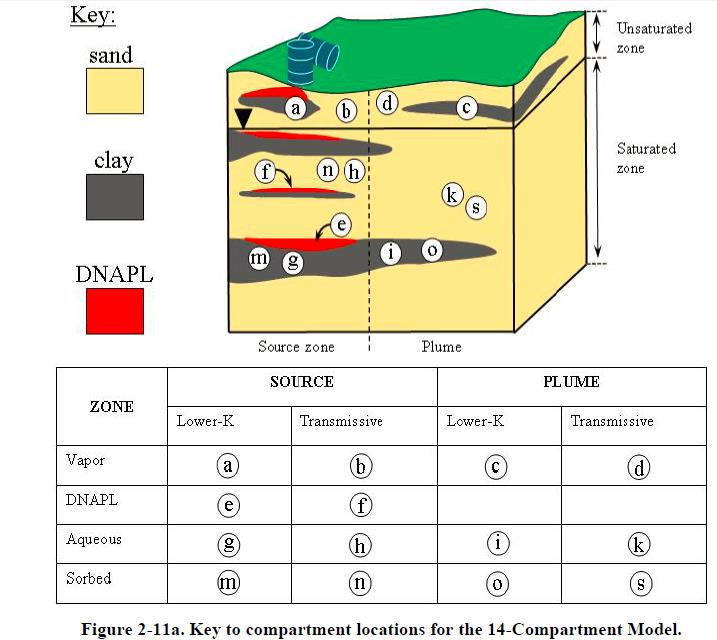
Table 2-1a shows a 14-Compartment Model representation in blank tabular form. One way to use the 14-Compartment Model is to fill in such a table with qualitative estimates of the chemical potential in each compartment. Then, mass transfers between compartments can be anticipated as contaminants flow from zones of higher chemical potential to zones of lower chemical potential. The transfers may be advective or diffusive. Table 2-1b shows the transfers that are possible among the various compartments.
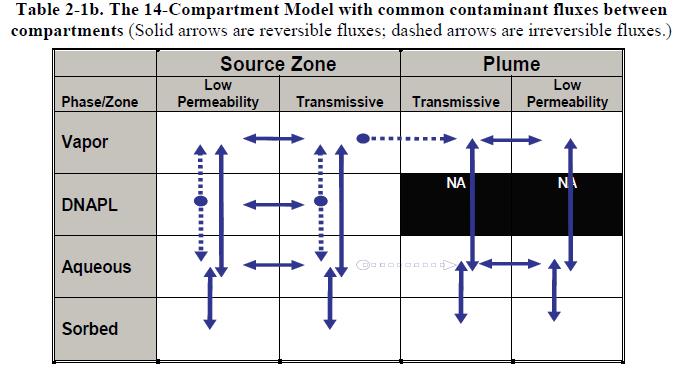
Table 2-2 fills in a table to represent the site situation shown in Figure 2-11a, just after the DNAPL spill occurred. At this stage, the chemical potential in the DNAPL phase is high, and there has been little mass transfer into other compartments. At this early stage of spill site maturation, the mass transfers are primarily from the DNAPL into other compartments.
In transmissive zones aqueous- and/or vapor-phase chlorinated solvents are carried with the flow of water or soil gas. In contrast, low-permeability zones are largely stagnant from a flow perspective. Critically, low-permeability zones store and release contaminants via diffusion (Freeze and Cherry 1979; Sudicky 1986; Parker, Gillham, and Cherry 1994; Chapman and Parker 2005; Sale et al. 2008).
As an introductory illustration, Figure 2-11b shows the 14-Compartment Model analysis of P&T in a source zone. P&T is usually considered an inefficient approach for depleting contaminants in source zones. However, since the technology recovers aqueous-phase chlorinated solvents from the transmissive portion of the source zone, a benefit of source zone P&T is reduced loading of aqueous-phase contaminants to transmissive zones in the downgradient plume.
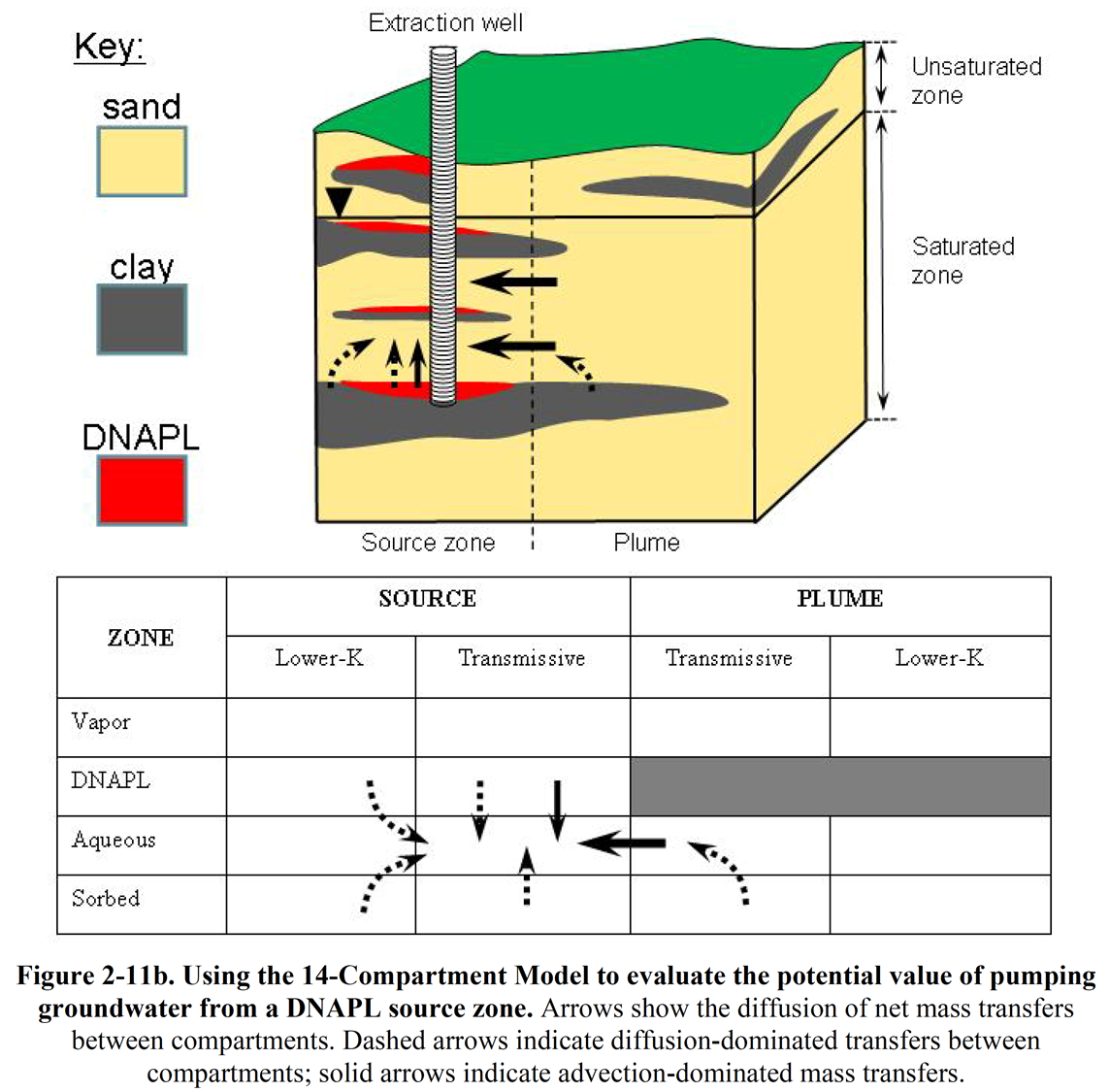
The following points provide an analysis of the mass transfers associated with groundwater extraction from a DNAPL source zone as depicted in Figure 2-11b:
- Pumping groundwater from the source will provide direct depletion of aqueous-phase contaminants in the transmissive portion of the source zone.
- If present, DNAPL will provide persistent release to the aqueous phase in the transmissive zone through dissolution.
- DNAPL may also be directly recoverable, especially during early-time pumping.
- Pumping groundwater from the source zone will provide direct depletion of aqueous-phase contamination from adjacent portions of the plume through advective mass transfer.
- Depletion of contamination in the transmissive zones results in the slow release of aqueous contaminants in lower-K zones, largely through diffusive mass transfer.
- Depletion of contamination in the transmissive zones also results in the slow release of sorbed-phase contaminants in lower-K zone, also through diffusive mass transfer. Note that the release of sorbed-phase contaminants can be a slow process.
Unfortunately, slow recovery of contaminants from adjacent compartments mandates long-term pumping.
2.5.2 Evaluating Overall Contaminant Distribution and Plume Maturity using the 14-Compartment Model
Table 2-3 provides a 14-Compartment Model illustration of the early, middle, and late stage plumes described in Section 2.4.1.
Table 2-3. Illustration of the progression of a DNAPL-sourced plume through time that results from mass transfers between compartments using the 14-Compartment Model (Groundwater flow [advection] carries contaminant from the source zone into the plume zone, and both diffusive and advective mass transfers can eventually distribute contaminants to all compartments. Over extended periods, contaminants can be diluted to lower concentrations, with stored mass in the lower-K zones acting as persistent sources of contamination.)
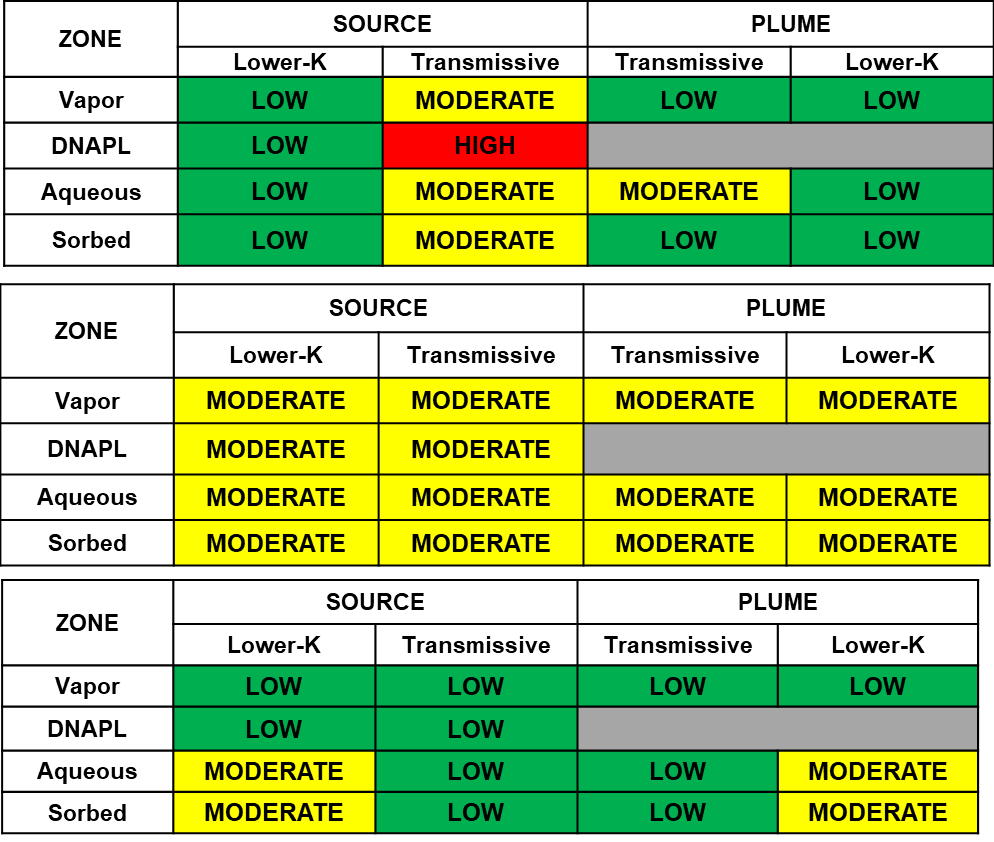
The transmissive zones of early stage plumes contain the highest VOC concentrations in their sources, particularity near the DNAPL phase. Over time the early-stage DNAPL phase, based on aqueous-phase equivalent concentration, is diminished by advection, biotic and abiotic degradation, and mass transfer into lower-permeability regions and other chemical phases within the source as well as the plume. In a middle stage, the aqueous-phase equivalent concentrations across affected phases and zones are relatively equal. In late-stage plumes, contaminant concentrations have attenuated in the more permeable (transmissive) zones, and the larger remaining concentrations remain in the lower-permeability zones within both the source and the plume. These concepts are a useful part of site-specific CSM development because plume maturity has a large effect on the response to treatment of a source and plume and therefore the potential efficacy of possible remediation efforts.
2.6 Application of the 14-Compartment Model to an Example Site
The following example is based on an actual site. It is used repeatedly in each chapter to follow to illustrate the application of the concepts and approaches introduced in each chapter supporting the development of an IDSS.
2.6.1 Characteristics of the Site
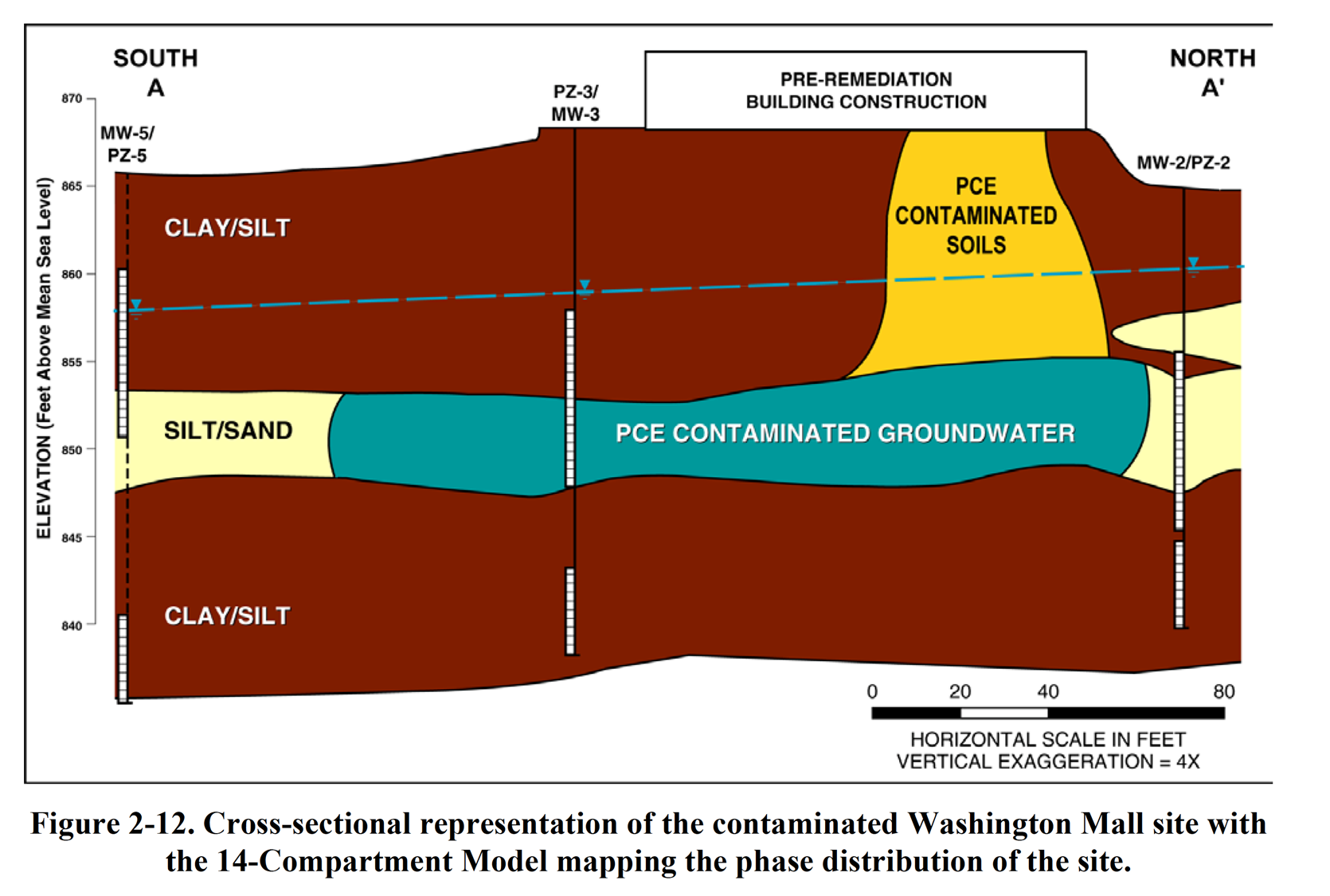
The Washington Square Mall was constructed in 1978. One tenant, a dry cleaner, released unknown quantities of PCE into the soils and subsequently into the underlying aquifer over a 2-year period. Approximately 130 tons of DNAPL containing soil was excavated and disposed as a hazardous waste in 1990. The community and state government are concerned that the property has fallen into disuse and is becoming dilapidated, with a 20% occupancy rate in 1999. A local developer is interested in redeveloping the property for commercial/retail use (i.e., its historic intended use) and is evaluating the life-cycle costs for remediating the site. The subsurface is composed of approximately 13 feet of clayey silt, overlying 6 feet of silty sand. Clayey silt below the silty sand zone extends at least another 10 feet below grade surface (bgs). Groundwater flows from the southwest to the northeast; depth to groundwater fluctuates 5–7 feet bgs. Residual PCE contamination has been measured in the soil at 4000 µg/kg and in groundwater at 180 µg/L near the point of release (the source area). There has been no remaining DNAPL confirmed/identified at the site; however, residual PCE continues to discharge into the underlying groundwater. PCE contamination has migrated approximately 200 feet downgradient of the point of release. There are no commercial buildings or residential homes downgradient of the site, and there are no potential adverse impacts to nearby drinking water wells.
2.6.2 14-Compartment Model Illustration
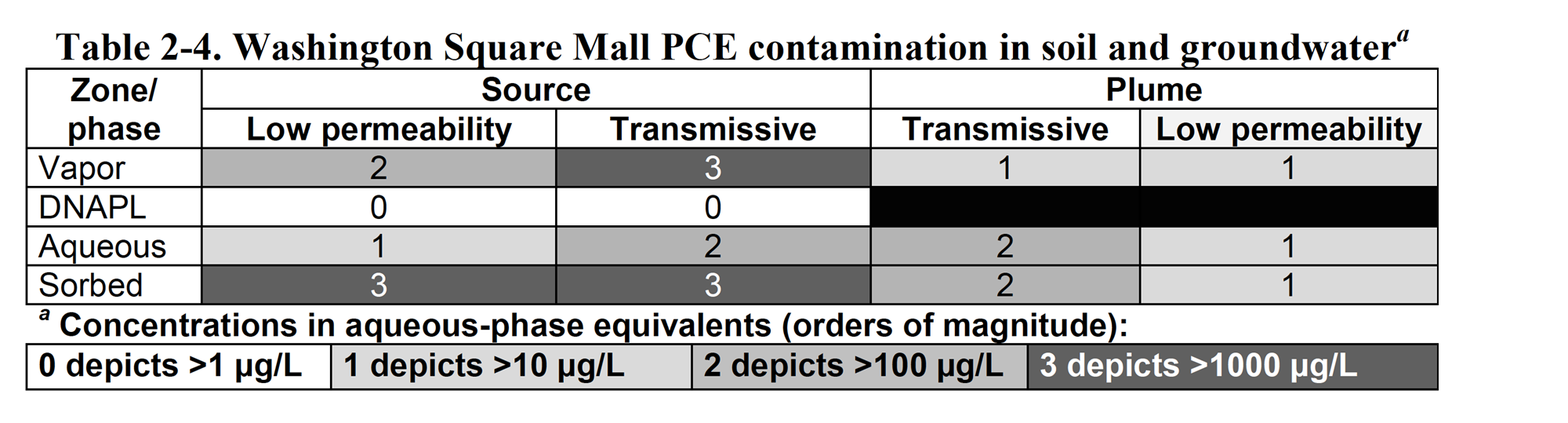
Figure 2-12 shows the CSM subsurface cross section integrated with the 14-Compartment Model. The 14-Compartment Model (Table 2-4) also depicts the order-of-magnitude distribution of contaminant concentrations. The concentration estimates in the aqueous and sorbed/residual cells in the source zone were developed from available water quality and soil gas data. The values in the transmissive zone of the plume were also developed from groundwater data. Concentrations in the other cells were derived as approximate expected values based on equilibrium partitioning between phases. Unfortunately, as is typical for many sites, no data were available from the low-permeability zones. When critical information is missing, efforts should be made to collect the information needed to make fully informed decisions. Thus, development of 14-Compartment Models can help decisions regarding collection of additional data. Overall, the Washington Square Mall resembles a middle-stage site (see Figure 2-8 and Sale and Newell 2011, Figure 11) where chlorinated solvent has apparently moved into the aqueous, vapor, and sorbed phases.
The establishment of achievable objectives is the next step in the process. The next chapter describes how to establish overarching objectives for the site and to reduce these to very specific, measurable, attainable, relevant, and time-bound (SMART) objectives.